Aufbau Diagram For Titanium. This video shows how to draw the orbital diagram of titanium ti. In its compounds titanium exhibits oxidation states of 2 3 and 4 as in the oxygen compounds titanium monoxide tio dititanium trioxide ti2o3 and titanium dioxide tio2 respectively.
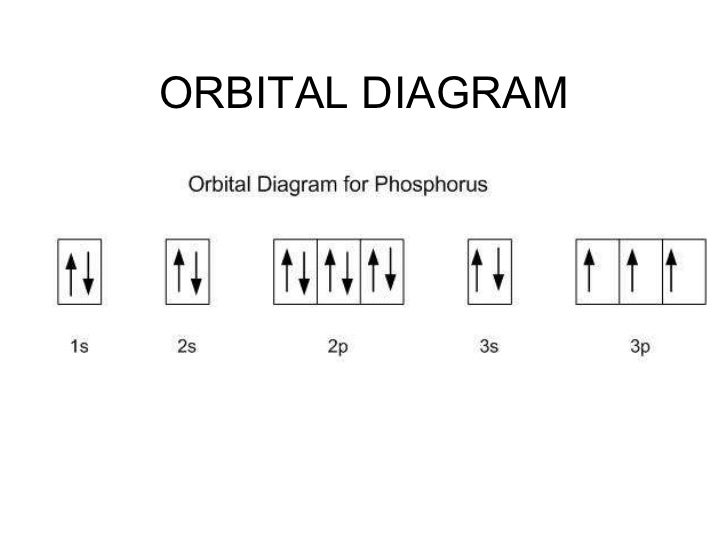
3p6 6 electrons giving a total of 18 which is the same as the noble gas argon. This video shows how to draw the orbital diagram of titanium ti. These energy levels contain sub shells or orbitals.
By contrast many compounds are formed by titanium in the 3 state.
For titanium you have 22 electrons to account for as follows. 2p6 6 electrons giving a total of 10. It is a mnemonic used to remember the order of filling of atomic orbitals during the construction of the ground state electron configurations of the elements. The electron configuration for titanium is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 2 4s 2 according to the jefferson lab website.
