Ch20 Lewis Structure. In order to complete the octets for all of the atoms in the structure you will need to a double bond between the carbon c and oxygen o atoms. How do you draw the lewis structure for ch2o.
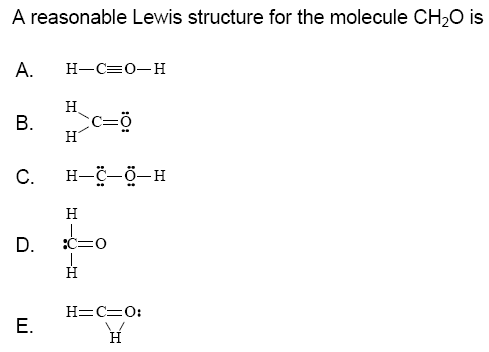
2 1 for each h 4 for c 6 for o 12. The approximate lewis structure of ch2o is this. The molecular geometry of ch 2 o is trigonal planar with asymmetric charge distribution.
In order to complete the octets for all of the atoms in the structure you will need to a double bond between the carbon c and oxygen o atoms.
Moreover the structure of ch2o is trigonal planar having the bond angles slightly distorted from the ideal percentage of 120. A step by step explanation of how to draw the ch2o lewis dot structure for the ch2o lewis structure calculate the total number of valence electrons for the. 2 1 for each h 4 for c 6 for o 12. How do you draw the lewis structure for ch2o.
