Ch2o Lewis Structure Hybridization. It is polar due to the difference in the partial charges on carbon and oxygen atom. It can be figured out with the help of the below mentioned formula.
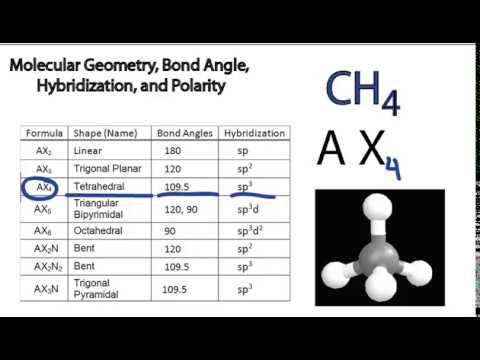
Total hybrid orbitals count of sigma bonds count of lone pairs on the central atom. Using the vsepr theory it can be seen that ch2o represents the chemical formula of ax3. In the case of a single bond there exists only one sigma bond.
And as per the vsepr rule compounds with ax3 have sp2 hybridization.
Total hybrid orbitals count of sigma bonds count of lone pairs on the central atom. Let s do the lewis structure for ch2o methanal or formaldehyde. Formaldehyde lewis structure ch2o electron geometry. Use google image search if you want to actually see the lewis diagram for pt.
