Cl2co Lewis Structure Polar Or Nonpolar. Learn to determine if cl2 is polar or nonpolar based on the lewis structure and the molecular geometry shape we start with the lewis structure and then use. What is polar and non polar.
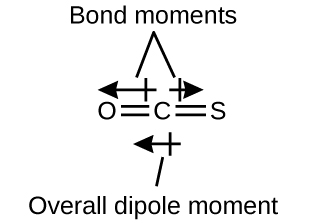
Draw the lewis structure. All of the following molecules you have listed are considered polar. I drew its lewis structure and got a trigonal planar shape with a double bond on the oxygen and i would normally think it was nonpolar because of the symmetrical shape however i am aware that o has a higher electronegativity than cl so maybe the net dipole moment might be pointing towards o which would make it polar but i m not sure.
I drew its lewis structure and got a trigonal planar shape with a double bond on the oxygen and i would normally think it was nonpolar because of the symmetrical shape however i am aware that o has a higher electronegativity than cl so maybe the net dipole moment might be pointing towards o which would make it polar but i m not sure.
Answer cl2co is polar. The lewis structure for cl 2 co requires you to place carbon in the center of the structure since it is the most electronegative. While the 2 cls have dipole moment components that partially cancel each other out there is a still a difference in dipole moment on the y. If5 is polar.
