Cs2 Lewis Structure 3d. So for the structure on the left the formal charge is. In lewis structure of cs2 molecule there are 16 valence electrons out of which four valence electrons are of carbon and six valence electrons are from each sulfur molecule.
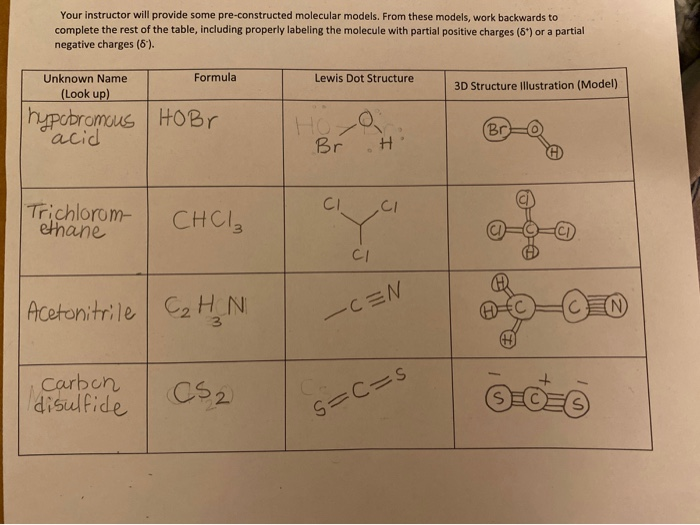
For the cs2 lewis structure calculate the total number of valence electrons for the cs2 molecule. These two sulfur molecules form double bonds with this carbon molecule to complete carbon s. Trithiocarbonate cs3 2 cid 16020005 structure chemical names physical and chemical properties classification patents literature biological activities.
These two sulfur molecules form double bonds with this carbon molecule to complete carbon s.
Therefore this molecule is nonpolar. Formal charge can be calculated in the following manner. The molecular geometry of cs 2 is linear with symmetric electron region distribution around the central atom. These structures are the best because they give the lowest formal charge and the structure with the formal charge closest to zero is the best choice.
