Draw The Electron Dot Structure Of Ethane Molecule C2h6. Draw the lewis structures for the following molecules and ions. Calculate the total valence electrons in the molecule.
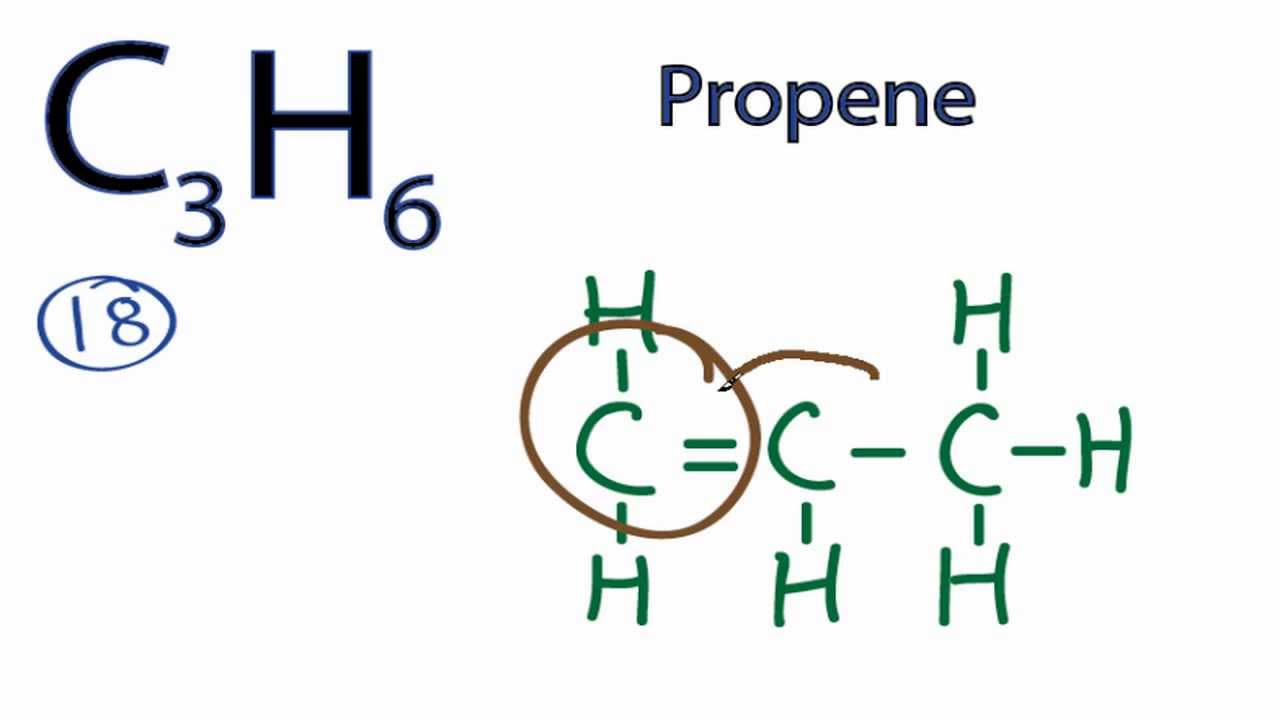
Write the electron dot structure of ethene molecule c 2. Ethane is an organic compound with a chemical formula of c2h6. This compound is one of the simplest hydrocarbons to exist having a single bond between carbon atoms.
Alternatively a dot method can be used to draw the lewis structure.
Alternatively a dot method can be used to draw the lewis structure. Find octet e for. It is a colorless and odorless molecule that exists as a gas at the standard room temperature. Write the electron dot structure of ethene molecule c 2.
