Electron Dot Structure Of Nh3 Class 10. Find how many electrons are required in total. Draw the electron dot structures for i ethanoic acid ii co2 iii f2 iv ch4.
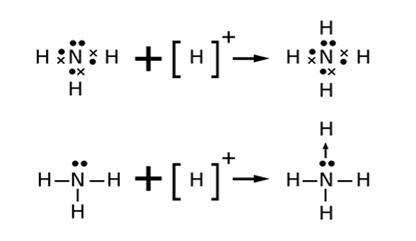
Lewis structures for nh3. Remember too that hydrogen only needs two valence electrons to have a full outer shell. This pair exerts repulsive forces on the bonding pairs of electrons.
To draw the lewis structure of nh3 first add the valence electrons.
For a molecule the lewis structure is the total valence electrons in the molecule. The shape is distorted because of the lone pairs of electrons. Draw the electron dot structure of n2 and nh3. Students upto class 10 2 preparing for all government exams cbse board exam icse board exam state board exam.
