Formaldehyde Lewis Structure Polar Or Nonpolar. Polar draw the lewis structure of ch f and then determine if the molecule is polar or nonpolar. Vsepr molecule shape molecular structure formaldehyde h 2 co or ch 2 o the difference is 3 44 polar.
It provides examples so you can quickly distinguish nonpolar molecul. Formaldehyde has two lone pairs of electrons on the oxygen atom and no lone pairs on the central atom. In any atom only those electrons found in the highest energy shell known as the valence shell take place in bonding.
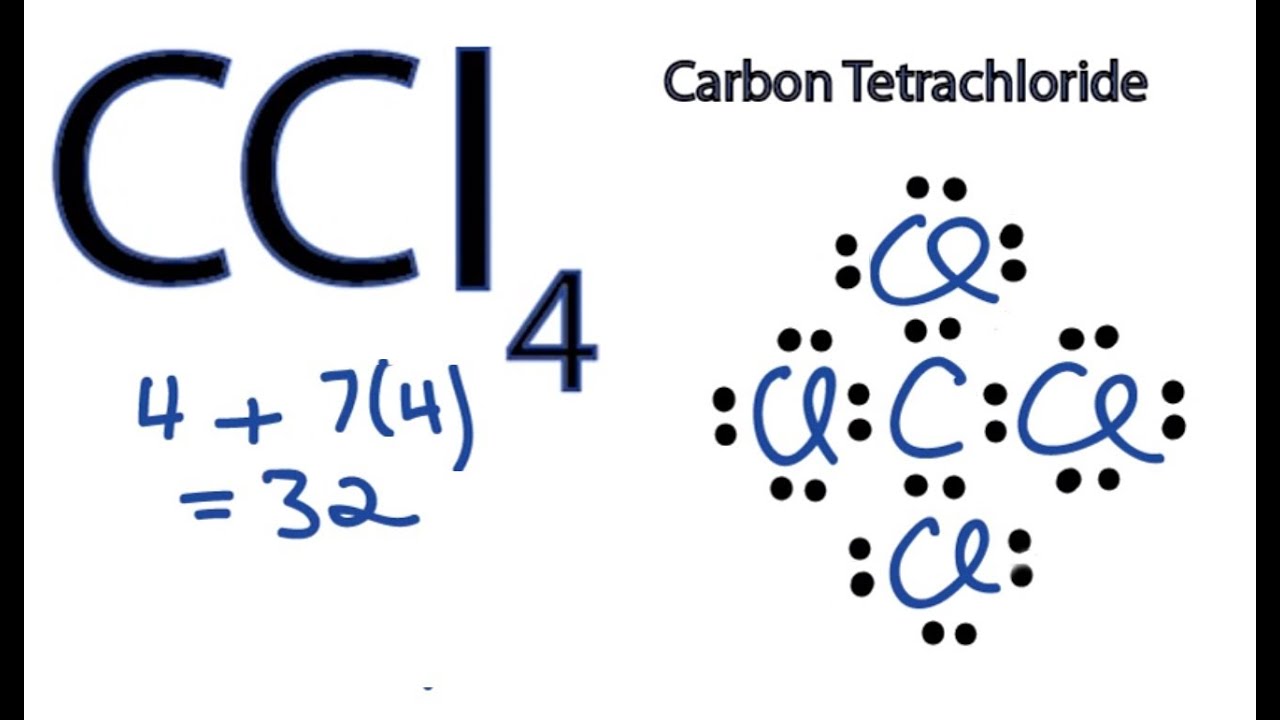
Calculate the total valence electrons in the molecule.
In any atom only those electrons found in the highest energy shell known as the valence shell take place in bonding. It is polar due to the difference in the partial charges on carbon and oxygen atom. Polar draw the lewis structure of ch f and then determine if the molecule is polar or nonpolar. Figure out the geometry using vsepr theory visualize or draw the geometry.
