Give The Electron Dot Structure Of Co2. Here are the steps that i follow when drawing a lewis structure. Asked aug 13 2019 in class x science by muskan15 3 443 points.
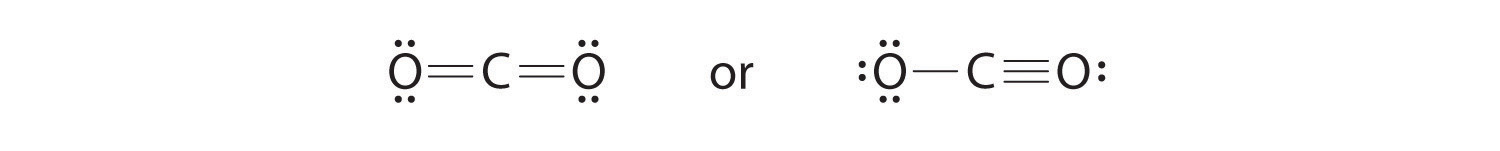
Here are the steps that i follow when drawing a lewis structure. I also go over hybridization shape and bond angles. Let s go over the lewis structure and find out how to interpret this representation of carbon dioxide.
Since there are only two oxygen atoms in an o 2 molecule the atoms form a double bond resulting in the following lewis electron dot structure.
Since there are only two oxygen atoms in an o 2 molecule the atoms form a double bond resulting in the following lewis electron dot structure. Dots can be used to represent the shared electrons within the bonds of the atoms but dashes can be used to represent covalent bonds as well. Here are the steps that i follow when drawing a lewis structure. Carbon dioxide chemical formula co2 is a chemical compound composed of two oxygen atoms covalently bonded to a single carbon atom.
