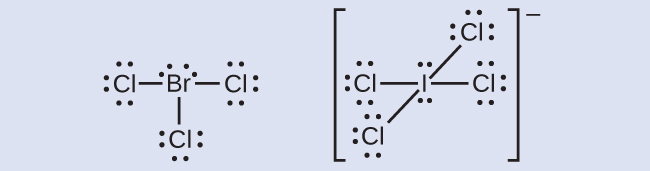
I2 Covalent Bond Lewis Structure. Hydrogen has 1 valence electron and iodine in group 7 with f and cl has 7 valence electrons. Hi is very similar to hf and hcl.
Single bonds have a bond order of 1 double bonds 2 and triple bonds 3. A plot of the overall energy of a covalent bond as a function of internuclear distance is identical to a plot of an ionic pair because both result from attractive and repulsive forces between charged entities. It is bonded to two of them by single covalent bonds and to the third by double bond.
Single bonds have a bond order of 1 double bonds 2 and triple bonds 3.
H f bond length 92 pm b. Hydrogen has 1 valence electron and iodine in group 7 with f and cl has 7 valence electrons. In lewis electron structures we encounter bonding pairs which are shared by two atoms and lone pairs which are not shared between. The bonds tie the atoms very tightly together into a layer.
