Lewis Structure Of Hno3 With Formal Charges. In this case the formal charges will be closer to zero if you place a double bond beteween the nitrogen atom and the oxygen atom without the h attached. Hon ho no oz.
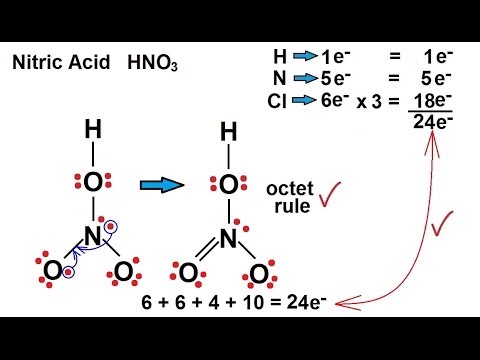
What is the correct lewis structure for nitric acid hno3 including the formal charges. Lewis structure of nitric acid there is a n o bond in nitric acid lewis structure. Hon ho no oz.
In the lewis structure of nitric acid there is a 1 charge on nitrogen atom and one double bond between nitrogen and one oxygen atom.
What is the correct lewis structure for nitric acid hno3 including the formal charges. The hno3 lewis structure is best thought of as the no3 with an h attache. A i b ii c iii d iv e none of these. Check the formal charges to be sure that each atom has a formal charge of zero.
