Lithium Carbonate Lewis Structure. One electron is transferred from lithium and makes it electro positive and by gaining one electron from lithium chlorine becomes electronegative. The lithium carbonate chemical formula is li 2 co 3 and its molar mass is 73 89 g mol 1 the molecule is formed by the lithium cation li carbonate anion co 3 2 and their crystal structure is monoclinic.
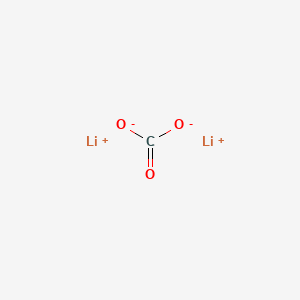
One electron is transferred from lithium and makes it electro positive and by gaining one electron from lithium chlorine becomes electronegative. I show you where lithium is on the periodic table and how to determine ho. Its chemical structure can be written as below in the common representations used for organic molecules.
Lithium carbonate usp 300 mg and is a two piece pink hard gelatin capsules size 2 imprinted with 300 on the body and g221 on the cap.
To understand how li interacts with individual carbonate molecules in nonaqueous electrolytes we conducted natural abundance 17o nmr measurements on electrolyte solutions of 1 m lipf6 in a series of binary solvent mixtures of ethylene carbonate ec and dimethyl carbonate dmc. Lithium carbonate is an inorganic compound the lithium salt of carbonate with the formula li 2 co 3 this white salt is widely used in the processing of metal oxides and treatment of mood disorders. To understand how li interacts with individual carbonate molecules in nonaqueous electrolytes we conducted natural abundance 17o nmr measurements on electrolyte solutions of 1 m lipf6 in a series of binary solvent mixtures of ethylene carbonate ec and dimethyl carbonate dmc. The lithium carbonate chemical formula is li 2 co 3 and its molar mass is 73 89 g mol 1 the molecule is formed by the lithium cation li carbonate anion co 3 2 and their crystal structure is monoclinic.
