Lithium Fluoride Bohr Diagram. Enthalpy of formation for f g 77 kj mol. It is a colorless solid that transitions to white with decreasing crystal size.
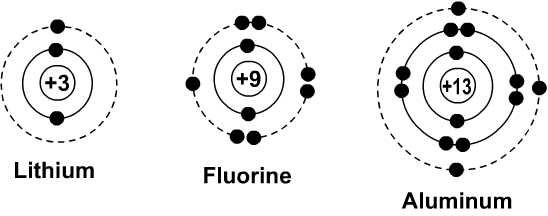
Atomization enthalpy of fluorine. F f bond dissocation energy 154 kj mol. Figure 2 contrast the bohr diagrams for lithium fluorine and aluminum atoms.
4 lirf 4 r y er yb lu rare earth doped.
Lithium yttrium fluoride liyf 4 ylf is the most popular fluoride laser crystal. Figure 7 the bohr diagrams for three of the group 2 elements a beryllium be b magnesium mg and c calcium ca. Electron affinity of fluorine. Calculate the lattice enthalpy for lithium fluoride given the following information.
