Lithium Ion Bohr Rutherford Diagram. Draw a bohr rutherford diagram for beryllium. Lithium oxygen calcium and phosphorus.
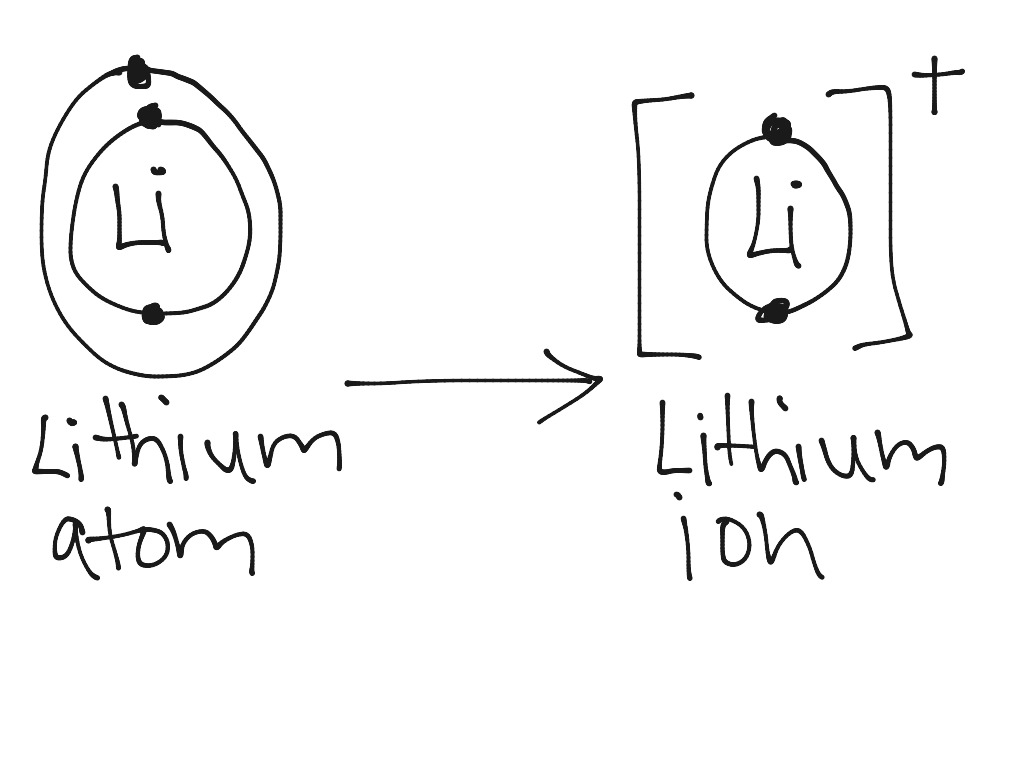
The shell closest to the nucleus is called the k shell next is the l shell next is the m shell. They all have one electron in their valence shell. Draw the following bohr model diagrams note they are ions be 2 cl f n3 ca2 k na o2 mg2 be2 s2 li chem ws 6 page 1.
Lithium oxygen calcium and phosphorus.
In atomic physics the bohr model or rutherford bohr model presented by niels bohr and ernest rutherford in 1913 is a system consisting of a small dense nucleus surrounded by orbiting electrons similar to the structure of the solar system but with attraction provided by electrostatic forces in place of gravity after the cubical model 1902 the plum pudding model 1904 the saturnian. When atoms form an ion describe the outer shell of that ion. Compare a sodium ion to a. Lithium oxygen calcium and phosphorus.
