Molecular Geometry Carbon Disulfide Lewis Structure. Therefore this molecule is nonpolar. In co2 lewis structure the carbon atom is in the central position as it is the least electronegative atom in the molecule.
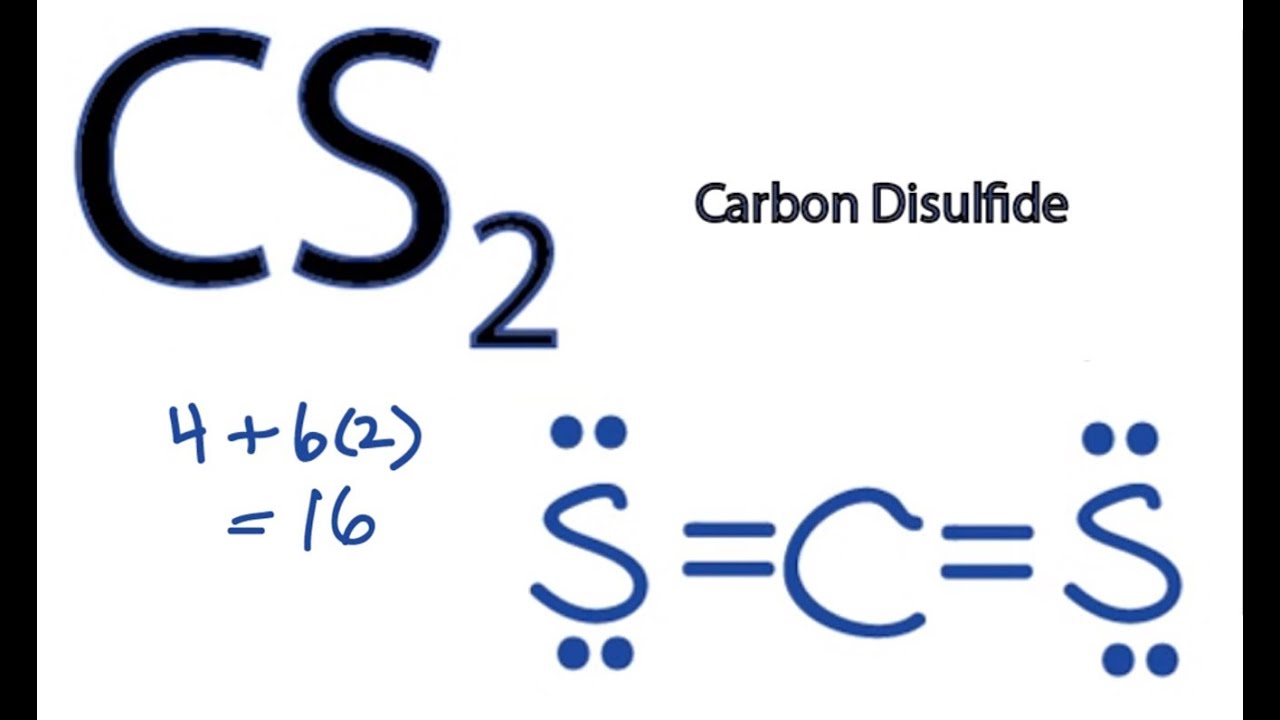
Carbon disulfide from the lewis structure above we can see that the central carbon atom has 2 electron domains also called negative charge centers. Therefore this molecule is nonpolar. We draw lewis structures to predict.
Cs2 is an abbreviated form of carbon disulphide.
The physical properties of a molecule such as boiling point surface tension etc. Lewis structure for cs 2 carbon disulfide lewis structure for cs. Next draw the 3d molecular geometry using vsepr rules. Two oxygen atoms are located on the terminals where.
