Molecular Orbital Diagram Of O2 2 Negative. The electronic configuration of c 2 is kk σ 2s 2 σ 2s 2 π 2p x 2 π 2p y 2. Experiments show that each o2 molecule has two unpaired electrons.
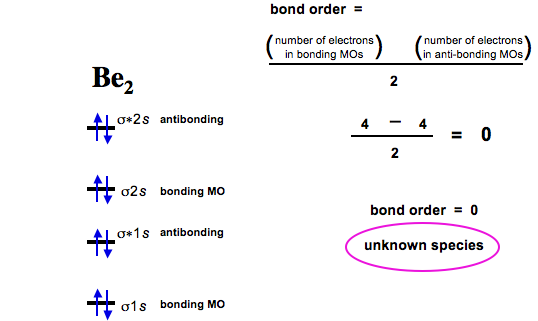
Bo 1 2 bonding e anti bonding e 1 2 8 4 2 lcao mo theory also predicts correctly that o2has two unpaired electrons. For a dioxygen negative 1 ion. Superoxides are compounds in which the oxidation number of oxygen is 1 2.
We illustrate how to use these points by constructing a molecular orbital energy level diagram for f 2 we use the diagram in part a in figure pageindex 1.
Molecular electron configuration for o2 σ2σ 2σ2π4π 2 we can also calculate the o o bond order. We can also notice the magnetic property of diamagnetic or paramagnetic. Atomic orbitals of oxygen combine to form molecular orbitals. We illustrate how to use these points by constructing a molecular orbital energy level diagram for f 2 we use the diagram in part a in figure pageindex 1.
