Molecular Shape Sif4 Lewis Structure. Alternatively a dot method can be used to draw the lewis structure. In this structure sulfur is the least electronegative element and so transfers in the middle of the structure and the diagram gives a three dimensional structural information.
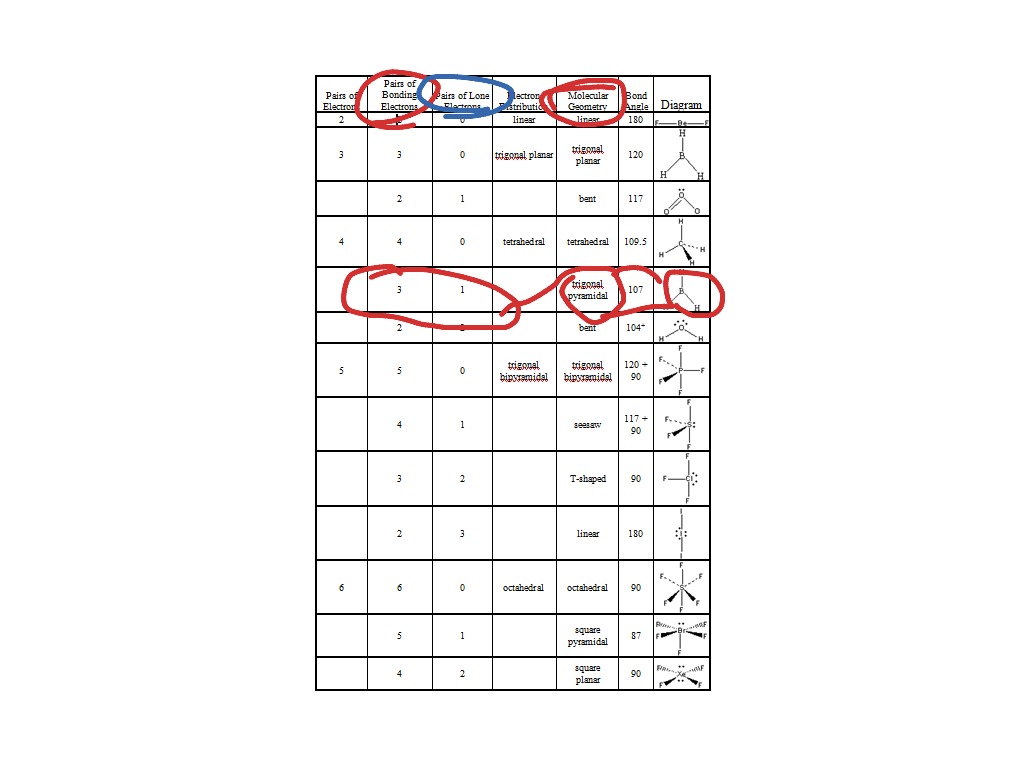
Krf4 square planar nonpolar. Sif4 molecule has a tetrahedral molecular geometry. In this structure sulfur is the least electronegative element and so transfers in the middle of the structure and the diagram gives a three dimensional structural information.
Chemistry learning made easy this tutorial will help you deal with the lewis structure and moleculargeometry for silane sih4.
In this structure sulfur is the least electronegative element and so transfers in the middle of the structure and the diagram gives a three dimensional structural information. In this structure sulfur is the least electronegative element and so transfers in the middle of the structure and the diagram gives a three dimensional structural information. Chemistry learning made easy this tutorial will help you deal with the lewis structure and moleculargeometry for silane sih4. This means that sef4 has trigonal bipyramidal structure with 4 bond pairs and 1 lone pair.
