Neon Ion Bohr Diagram. Bohr diagrams indicate how many electrons fill each principal shell. Draw a bohr rutherford diagram of a sodium atom.
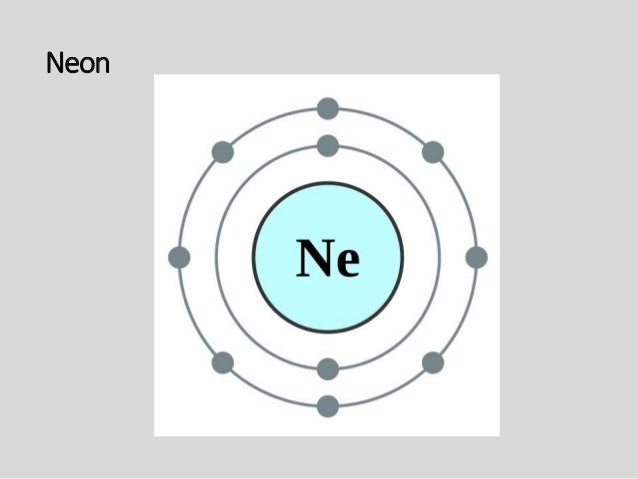
Group 18 elements helium neon and argon are shown have a full outer or valence shell. Bohr diagrams indicate how many electrons fill each principal shell. The third orbital contains eight again except that in the good question.
The bohr model for neon shows a nucleus with ten protons and neutrons with ten electrons orbiting the nucleus in two different energy levels.
Two electron shells surrounding the nucleus containing 2 electrons in the n 1 shell and 8 electrons in the n 2 shell. Neon has 2 electrons in its first shell and 8 in its secondcheck me out. Group 18 elements helium neon and argon are shown have a full outer or valence shell. Bohr s model of the atom.
