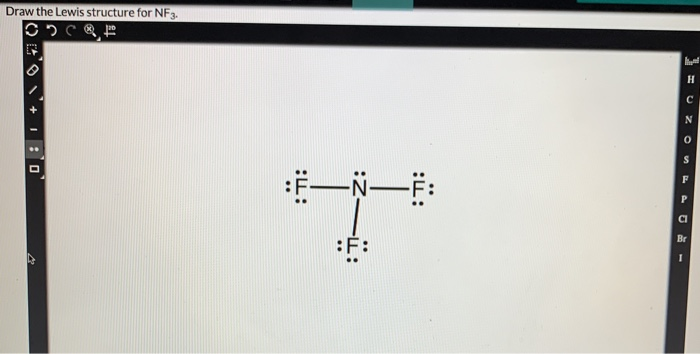
Nf3 Lewis Structure Molecular Geometry. The nf3 is polar because they distribute bonding electrons and its shape also balances the polarity. Bf3 has planar shape while nh3 has pyramidal because b has 3 electrons in the outermost shell while n has 5 elctrons in its outermost shell.
Bf3 has planar shape while nh3 has pyramidal because b has 3 electrons in the outermost shell while n has 5 elctrons in its outermost shell. The shape is distorted because of the lone pairs of electrons. Lewis structure of nf3 w free video guide.
Put nitrogen in center and arrange fluorine atoms on the sides.
Draw the lewis structure indicate the molecular structure. The nf3 is polar because nf3 has a lewis structure and the molecules form in a pyramidal shape. The shape is distorted because of the lone pairs of electrons. Additionally is bf3 trigonal pyramidal.
