Ozone Lewis Structure Polarity. It is an allotrope of oxygen that is much less stable than the diatomic allotrope o 2 breaking down in the lower atmosphere to o 2 ozone is formed from dioxygen by the action of ultraviolet uv light and electrical discharges within the. O3 is a polar molecule and it is due to its bent molecular geometry.
112 that has a characteristic pleasant odour it is continuously produced in the upper atmosphere by the action of solar ultraviolet radiation on atmospheric oxygen it is an antimicrobial agent used in the production of bottled water as well as in the treatment of meat poultry and other foodstuffs. An explosive pale blue gas b p. Let s study this in detail.
Let s study this in detail.
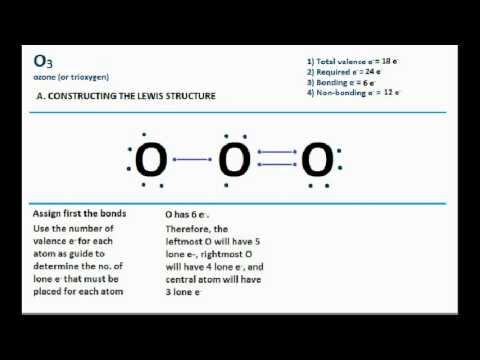
112 that has a characteristic pleasant odour it is continuously produced in the upper atmosphere by the action of solar ultraviolet radiation on atmospheric oxygen it is an antimicrobial agent used in the production of bottled water as well as in the treatment of meat poultry and other foodstuffs. Lewis structures a lewis structure shows how the valence electrons are arranged and indicates the bonding. Molecular structure of o3. An explosive pale blue gas b p.
