Sf4 Lewis Structure Bond Angle. Sf4 sulfur tetrafluoride sulfur tetrafluoride has 5 regions of electron density around the central sulfur atom 4 bonds and one lone pair. Sf4 bond angles and shape the central sulfur atom forms four bonds with the neighboring fluorine atoms and has one lone pair of electrons.
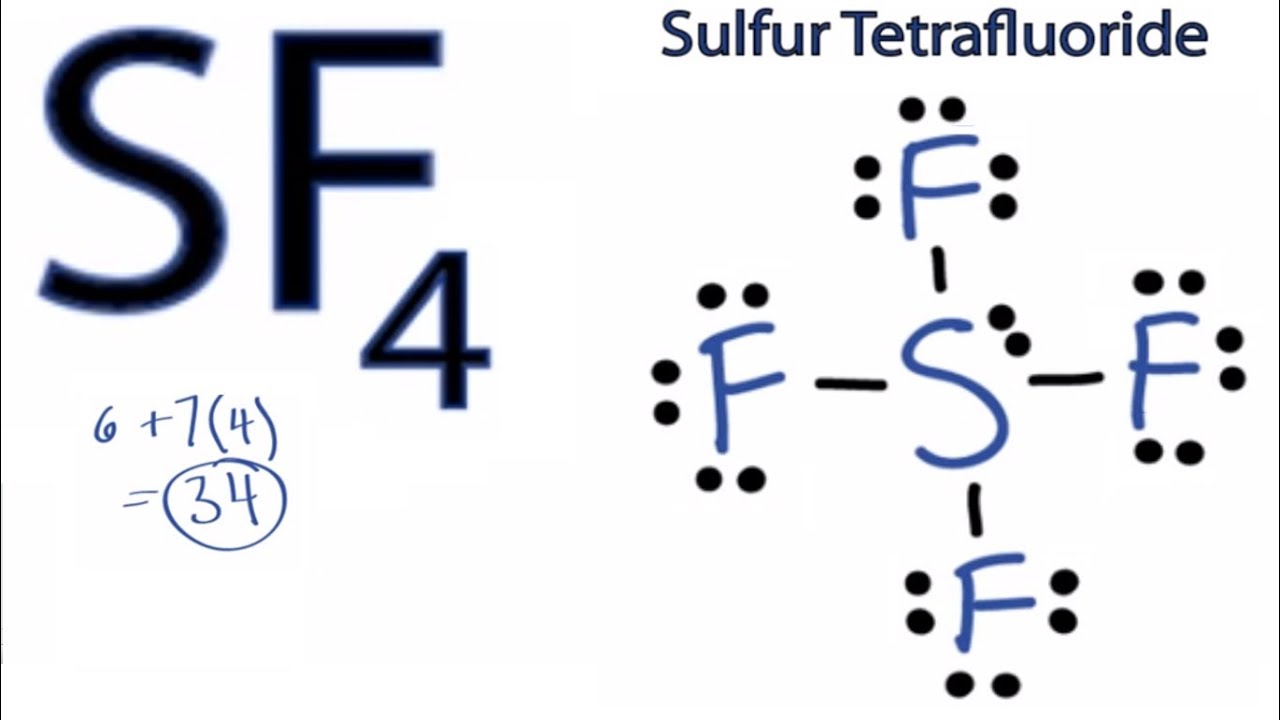
Sf4 bond angles and shape the central sulfur atom forms four bonds with the neighboring fluorine atoms and has one lone pair of electrons. One of the three equatorial positions is occupied by a nonbonding lone pair of electrons. I also go over formal charge hybridization shape and bond angle.
These are arranged in a trigonal bipyramidal shape with 102 f s f bond angles between the equatorial fluorine atoms and 173 between the axial fluorine atoms.
The structure of sf4 can therefore be anticipated using the principles of vsepr theory. Click to see full answer. One of the three equatorial positions is occupied by a nonbonding lone pair of electrons. The axial fluorine atom angel is 173 instead of the actual 180 o bond angle.
