Silicon Tetrafluoride Sif4 Lewis Structure. Five alarm fronts and leatherworks. Average rating 0 5.
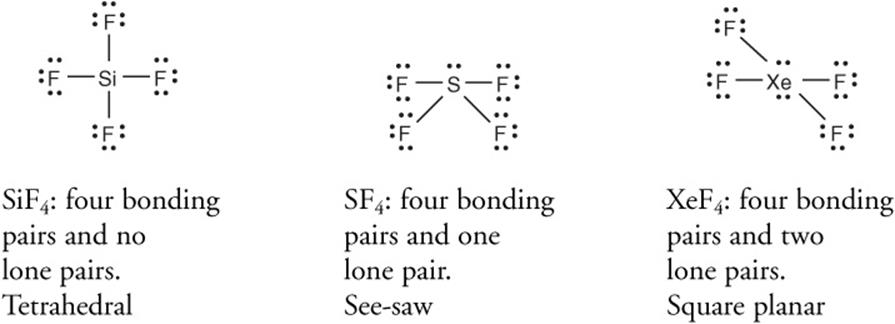
Substrate structure controls substitution mechanism s n 1 or s n 2. It is a tetrahedral molecule. The lewis structure of sf4 is the combination of 34 valence electron and 5 electron pairs around the sulfur in which there are four bonding pairs and one lone pair.
It was first synthesized by john davy in 1812.
It was first synthesized by john davy in 1812. Its boiling point is only 4 c above its melting point. Be the first to rate this page. It is a tetrahedral molecule.
